Alzheimer’s disease (AD) diagnosis in early stages is an important step that helps neurologists treat the patient in a timely manner and can lead to excellent outcomes in patient response to treatment plans. An ensemble-based machine learning framework presented, which is based on magnetic resonance imaging (MRI) attributes, can enhance AD early detection and diagnosis accuracy. Several classifiers are studied, including random forest, gradient boosting, and voting classifiers. The article will develop a framework of an ensemble model of these classifiers that can lead to enhanced accuracy of Alzheimer’s disease diagnosis. Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset is used in this research, and the MRI features are preprocessed and then implemented using the proposed model. The research results show a better performance of the ensemble models in terms of accuracy, precision, and recall, compared to individual classifiers. The effective integration of different classifiers to capture MRI patterns and the establishment of a strong diagnostic model with the possibility of being embedded within clinical decision support systems are the key contributions of this research. These softwares could be used by different stakeholders, like neurologists, patients. This may revolutionize traditional diagnostic and treatment methods by providing accurate and effective early diagnosis and treatment before the onset of Alzheimer’s disease.
Keywords: Alzheimer’s Disease Diagnosis, Machine learning, Ensemble learning, Magnetic resonance imaging, Classification Algorithms, Decision Support Systems
I. INTRODUCTION
Alzheimer’s disease (AD) ranks among the utmost devastating neurodegenerative disorders, disturbing millions globally and imposing major socioeconomic and emotional problems. Specified by continuous memory damage, cognitive decline, and behavioral changes, AD not only weakens individual independence, but also stresses caregivers and healthcare systems. The global occurrence of Alzheimer’s continues to rise, driven by old populations, increasing the urgency to address the diagnosis and treatment challenges [1].
Early diagnosis of Alzheimer’s disease is important for improving AD patient life. Identifying the disease in its early stages can help with intervention plans to slow progression and improve quality of life. This early identification also enables patients and families to plan health care treatments, accessing support services. Despite these benefits, current diagnostic methods often diagnose AD only after significant neurological damage has occurred, limiting therapeutic success [2].
Traditional AD diagnosis depends mainly on clinical, neuropsychological tests, and standard imaging methods. Magnetic resonance imaging (MRI) provides detailed body information, yet human interpretation may miss the early diagnosis of AD. Additionally, current diagnostic machines do not have integration with computational approaches that can attach complex MRI data, showing patterns unseen to the human eye [3].
This research aims to find a solution for these gaps by implementing a complete ensemble machine learning (ML) framework for the diagnosis of Alzheimer’s disease in its early stages, using MRI features. By joining multiple classifier algorithms—random forest, gradient boosting, and voting classifiers—into a unified ensemble model, the framework pursues to enhance accuracy, precision, and recall compared to a single algorithm. We used the Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset for building the proposed model. The final research goal is to develop a robust data-driven model adaptable to clinical decision support systems to improve early AD diagnosis, and eventually helping stakeholders, such as neurologists, patients, and the larger healthcare domain.
This paper is organized as follows: Section 2 outlines the background and reviews work already conducted on the early diagnosis of AD. Section 3 discusses the proposed ensemble model design including the data sets and pre-processing. In Section 4 we present the conclusion and future works.
II. LITERATURE REVIEW
A. Background
Alzheimer’s disease, which is characterized by a progressive decline in memory and cognitive function, is the most widespread form of dementia and is one of the major causes of mortality among the elderly [4], [5]. Even with the distinguished research efforts to develop disease-modifying treatments, AD remains without a definitive cure [6]. Therefore, detection of the early onset of AD requires sensitive approaches that can detect brain changes before reaching a point where the damage is permanent.
Magnetic resonance imaging (MRI) represents a vital tool in AD diagnosis, due to its high-resolution visualization of the anatomical structure of the brain. The challenge lies in the classic handling and interpretation of these images, which needs improvement in terms of sensitivity and reliability [3]. Machine learning (ML) has significantly evolved the interpretation of MRI images, due to its ability to detect features and manifestations that may not be observed by clinicians [7], [2]. Utilizing MRI data, ML techniques were used for the detection, classification, and prediction of AD. Ensemble learning models, which are the result of combining different classifiers and leverage the strengths of separate models to deliver better performance and superior accuracy.
B. Recent Research Works
Several studies explored the use of ensemble models for AD detection and classification; one study introduced a deep learning-based ensemble model for the early detection of AD using MRI images from ADNI dataset. This model was composed of six convolutional neural networks (CNN) and showed promising results [2].
Another study presented an ensemble deep learning model for the detection of AD; the paper presented two low-parameter CNNs: IR-BRAINNET and Modified-DEMNET [8].
Alruily et al. developed a unique deep learning ensemble model that fuses features from VGG16, MobileNet, and InceptionResNetV2, which aimed to increase AD diagnosis accuracy [9].
Nguyen et al. presented an ensemble learning model composed of both deep learning and machine learning, which achieved an approximate scoring time of 10 minutes, much faster than feature extraction-based methods [10].
A study in 2022 presented an ensemble model for AD diagnosis and made a comparison between their model and other machine learning models. In terms of accuracy, the presented model showed better results than previous models [7].
Shaffi et al. constructed a ML-based ensemble model that uses ADNI and OASIS datasets for the classification of AD. After putting ML models against DL models and comparing them in terms of performance, the study concluded that ML models surpassed DL models in terms of performance [3], while a study in 2021 utilized Diffusion Tensor Imaging (DTI) measures to construct an ensemble learning model that helps differentiate patients with AD [11].
A study in 2024 also presented an ensemble deep learning model, but based on quantum machine learning for AD classification. The model had 99.89% accuracy and is said to have superior accuracy and training time when compared to Support Vector Machine (SVM) [12].
III. PROPOSED MODEL AND METHODOLOGICAL FRAMEWORK
A. Dataset
The Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset is a very important clinical dataset that can help in proceeding Alzheimer’s research. It includes groups of MRI scans, demographic, and clinical tests that can be used for machine learning (ML) to implement early AD detection models. This study uses MRI features, a subset of the ADNI dataset, centering on the most impactful features that can help diagnose AD in its early stages.
B. Feature Extraction
Extracting meaningful features from MRI data is critical for effective classification. We utilize the following MRI features: Ventricles, Hippocampus, WholeBrain, Entorhinal, Fusiform, MidTemp, ICV. A preprocessing stage is applied to MRI features, employing imputation using the Multiple Imputation by Chained Equations (MICE) technique, and feature selection using Principal Component Analysis (PCA), then serving as inputs for the ensemble machine learning classifier.
C. Ensemble Model Development
Three machine learning algorithms support the classification framework: random forest, gradient boosting, and voting classifiers. Random forest works through a collection of decision trees, each one trained using bootstrap, that can provide solid results with respect to working with categorical feature communications and overfitting [13]. Gradient boosting revises weak classifiers by decreasing classification errors to the minimum, reaching detailed predictive accuracy, mainly with heterogeneous features [14]. The voting classifier constructs classifications from different independent models, depending on either weighted votes or majority, which can balance the variances and biases of each single model to produce an agreement diagnosis [15]. We have chosen these classifiers because of their excellent strengths reached in past AD classification researches.
The ensemble framework combines the three classifiers into an integrated model that increases their collective classification power. Implementation consists of training each classifier alone on the MRI features from ADNI datasets, then combining their classification outputs using a strict voting plan where the majority voting dominates the final classification class. This process framework schema is drawn showing parallel classifiers converging into an ensemble classification node as shown in Fig. 1. Model development experiments were implemented in SPSS and Python programming languages using an Intel Core i7 CPU with 64 GB RAM. The experimental setup needs a stratified 10-fold cross-validation to keep class distribution and alleviate overfitting. This integrative framework enhances classification accuracy by alleviating each classifier’s weaknesses and utilizing various algorithms’ strengths as a group [2], [3].
The ensemble framework methodology offers real benefits with respect to individual classifiers in AD diagnosis. Mainly, it reaches better classification accuracy, precision, and recall by combining various predictive views, reducing tendency to single-classifier errors and overfitting problems. Empirical studies affirm that ensemble classifiers give better robustness with the heterogeneity of clinical MRI scans data. Furthermore, the data-driven model can be integrated into decision support systems or software systems, providing neurologists with robust diagnostic tools that increase clinical decisions. The integration with real-time application in clinical diagnosis workflows and personalized treatment marks a revolution in computational neurodiagnostics diagnosis [16], [2]. The demonstrated efficiency and interactions of ensemble classification models confirm the potential of transforming traditional diagnostic models in Alzheimer’s disease management.
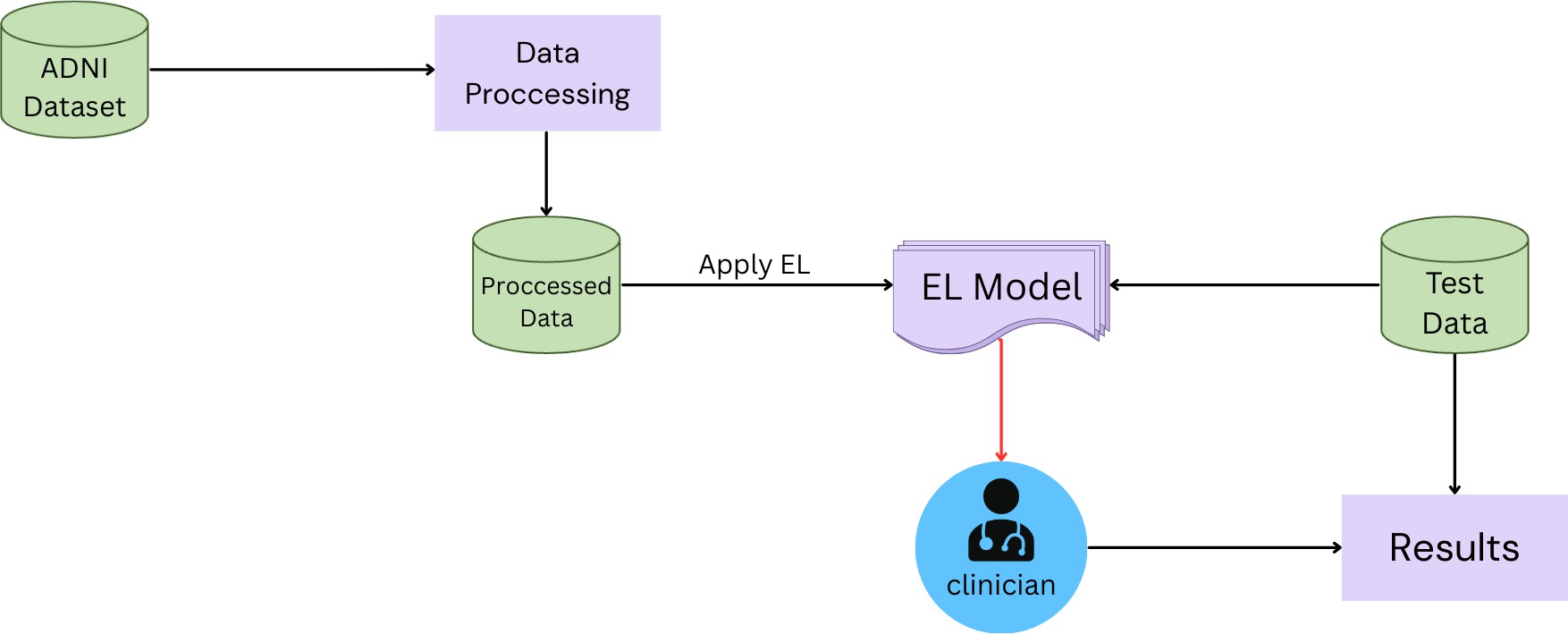 Fig. 1. Ensemble learning classification model
Fig. 1. Ensemble learning classification model
IV. CONCLUSION AND FUTURE WORKS
The ensemble model framework implemented in this research joins random forest, gradient boosting, and voting classifiers to exploit the strength of magnetic resonance imaging (MRI) features for Alzheimer’s disease diagnosis. The main characteristics driving its performance include the ability to manage complex, non-linear patterns and to reduce bias and variance by consolidating multiple model outputs. This integration produces a more accurate and robust classification model that surpasses individual classifiers in the diagnosis of Alzheimer’s disease.
For neurologists, the developed model provides an effective increase in diagnosis of Alzheimer’s Disease. Patients can benefit from early diagnosis, which can slow the progression and improve quality of life. Healthcare schemes may take advantage of reducing continuing burdens by enabling prior resource allocation, targeted treatments, and improving patient monitoring. Thus, the ensemble framework aligns well to develop decision support systems that help in early diagnosis and treatment of the growing AD rate.
Future research aimed at refining and implementing the ensemble model in routine treatment and clinical work will require integration with real-time existing decision support systems. Future recommended steps need constructing pipelines for MRI feature preprocessing, validating ensemble model outputs within diverse medical populations, and implementing user-friendly interfaces for neurologists to perform automated diagnostics.
The limitation is that the research dependence on MRI features excludes functional MRI or PET imaging, which could expose additional biomarkers vital for the early stage diagnosis of AD [16].
REFERENCES
[1] Givian, H., Calbimonte, J. P., & for the Alzheimer’s Disease Neuroimaging Initiative. (2024). Early diagnosis of Alzheimer’s disease and mild cognitive impairment using MRI analysis and machine learning algorithms. Discover Applied Sciences, 7(1), 27.
[2] Fathi, S., Ahmadi, A., Dehnad, A., Almasi-Dooghaee, M., Sadegh, M., & Alzheimer’s Disease Neuroimaging Initiative. (2024). A deep learning-based ensemble method for early diagnosis of Alzheimer’s disease using MRI images. Neuroinformatics, 22(1), 89–105.
[3] Shaffi, N., Subramanian, K., Vimbi, V., Hajamohideen, F., Abdesselam, A., & Mahmud, M. (2024). Performance evaluation of deep, shallow and ensemble machine learning methods for the automated classification of Alzheimer’s disease. International Journal of Neural Systems, 34(07), 2450029.
[4] Janghel, R. R., & Rathore, Y. K. (2021). Deep convolution neural network based system for early diagnosis of Alzheimer's disease. Irbm, 42(4), 258–267.
[5] An, X., Zhou, Y., Di, Y., & Ming, D. (2020, November). Dynamic functional connectivity and graph convolution network for Alzheimer's disease classification. In Proceedings of the 2020 7th International Conference on Biomedical and Bioinformatics Engineering (pp. 1–4).
[6] Huang, L., Yang, H., Che, Y., & Yang, J. (2024). Automatic speech analysis for detecting cognitive decline of older adults. Frontiers in Public Health, 12, 1417966.
[7] Khan, Y. F., Kaushik, B., Chowdhary, C. L., & Srivastava, G. (2022). Ensemble model for diagnostic classification of Alzheimer’s disease based on brain anatomical magnetic resonance imaging. Diagnostics, 12(12), 3193.
[8] Naderi, M., Rastgarpour, M., & Takhsha, A. R. (2024). Early Diagnosis of Alzheimer's Diseases and Dementia from MRI Images Using an Ensemble Deep Learning. arXiv preprint arXiv:2412.05666.
[9] Alruily, M., Abd El-Aziz, A. A., Mostafa, A. M., Ezz, M., Mostafa, E., Alsayat, A., & El-Ghany, S. A. (2025). Ensemble deep learning for Alzheimer’s disease diagnosis using MRI: Integrating features from VGG16, MobileNet, and InceptionResNetV2 models. PLOS ONE, 20(4), e0318620.
[10] Nguyen, D., Nguyen, H., Ong, H., Le, H., Ha, H., Duc, N. T., & Ngo, H. T. (2022). Ensemble learning using traditional machine learning and deep neural network for diagnosis of Alzheimer’s disease. IBRO Neuroscience Reports, 13, 255–263.
[11] Lella, E., Pazienza, A., Lofu, D., Anglani, R., & Vitulano, F. (2021). An ensemble learning approach based on diffusion tensor imaging measures for Alzheimer’s disease classification. Electronics, 10(3), 249.
[12] Jenber Belay, A., Walle, Y. M., & Haile, M. B. (2024). Deep ensemble learning and quantum machine learning approach for Alzheimer’s disease detection. Scientific Reports, 14(1), 14196.
[13] Sarica, A., Cerasa, A., & Quattrone, A. (2017). Random forest algorithm for the classification of neuroimaging data in Alzheimer's disease: a systematic review. Frontiers in Aging Neuroscience, 9, 329.
[14] Shojaie, M., Cabrerizo, M., DeKosky, S. T., Vaillancourt, D. E., Loewenstein, D., Duara, R., & Adjouadi, M. (2022). A transfer learning approach based on gradient boosting machine for diagnosis of Alzheimer’s disease. Frontiers in Aging Neuroscience, 14, 966883.
[15] Chatterjee, S., & Byun, Y. C. (2022). Voting ensemble approach for enhancing Alzheimer’s disease classification. Sensors, 22(19), 7661.
[16] Devi, B. M., & Ganesh, D. (2024, November). Brain Image Analysis for Alzheimer’s Disease Detection using Ensemble Machine Learning. In 2024 8th International Conference on Electronics, Communication and Aerospace Technology (ICECA) (pp. 779–785). IEEE.
